14 Terms
14 TermsHome > Terms > English, UK (UE) > IRB
IRB
Institutional Review Board. 1. A committee of physicians, statisticians, researchers, community advocates, and others that ensures that a clinical trial is ethical and that the rights of study participants are protected. All clinical trials in the U.S. must be approved by an IRB before they begin. 2. Every institution that conducts or supports biomedical or behavioural research involving human participants must, by federal regulation, have an IRB that initially approves and periodically reviews the research in order to protect the rights of human participants.
This is auto-generated content. You can help to improve it.
0
0
Improve it
- Part of Speech: noun
- Synonym(s):
- Blossary:
- Industry/Domain: Pharmaceutical
- Category: Clinical trials
- Company:
- Product:
- Acronym-Abbreviation:
Other Languages:
Member comments
Terms in the News
Featured Terms
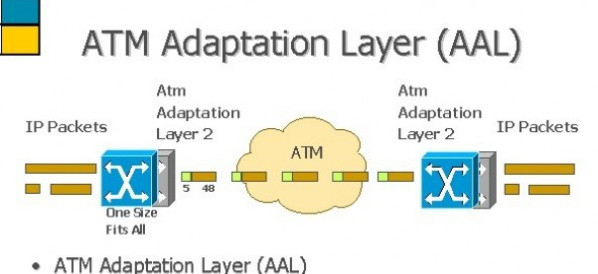
Industry/Domain: Telecommunications Category: General telecom
Atm adaptation layer
The ATM Adaptation Layer (AAL) is designed to support different types of applications and different types of traffic, such as voice, video, imagery, ...
Contributor
Featured blossaries
Browers Terms By Category
- Physical geography(2496)
- Geography(671)
- Cities & towns(554)
- Countries & Territories(515)
- Capitals(283)
- Human geography(103)
Geography(4630) Terms
- Air conditioners(327)
- Water heaters(114)
- Washing machines & dryers(69)
- Vacuum cleaners(64)
- Coffee makers(41)
- Cooking appliances(5)
Household appliances(624) Terms
- Poker(470)
- Chess(315)
- Bingo(205)
- Consoles(165)
- Computer games(126)
- Gaming accessories(9)
Games(1301) Terms
- Industrial automation(1051)
Automation(1051) Terms
- General furniture(461)
- Oriental rugs(322)
- Bedding(69)
- Curtains(52)
- Carpets(40)
- Chinese antique furniture(36)